2017 DMUC Key Discussion Highlights & Action Items
CMDP Training (Monday, 5/8 am and Thursday 5/11 am)
Feedback & Discussion:
User Defined Fields
User defined fields for CMDP came up in the context of sample point management (ex. By a school). In CMDP, there are user comment fields that could serve as a user defined field. CMDP does not support creation of customized data fields.
Adding Additional Analytes to Dropdown Menus
During primacy agency transition, the CMDP team is engaging State CMDP Administrators to determine if there are analytes that are regulated at the primacy agency level. As long as the reference data for the analyte conforms to the CMDP database schema, the state can add state-specific regulated analytes. The state would have to make these data available as part of its transition process. For example, the analyte name, analyte code, result UOM, method detection limit, detection limit UOM, applicable method name(s), applicable method codes, etc. These state-specific analytes will be stored in the reference tables and available to that primacy agency and its submitters.
Lag time / Effective dates
Lag times and effective dates are determined by the compliance determination process not the CMDP.
Request to include a box to mark “yes” or “no” for compliance
The CMDP’s primary purpose is to support laboratory and water systems electronic reporting of drinking water samples to primacy agencies All compliance determinations are done by SDWIS State or by Prime.
Composite Sample Migration to SDWIS State via DSE?
This requirement has been noted and added to the CMDP enhancement list for development.
Visually Indicate if Specific Validation Data is Available
No user interface design changes are currently planned for the CMDP. This enhancement idea could be implemented in the future.
How Does CMDP Resubmission Work to Correct Errors in the Validation Report?
The submitter has two options for correcting before e-signature:
- Delete the entire sample Job (all Sample IDs even those without errors), or only the affected Sample IDs (within a Job).
- Don’t delete anything. Use the Data Validation Report to identify which Sample ID has the error and then make the correction directly in CMDP using the web forms (for any submittal, everything is rendered as web forms so the user may see all the details).
If the submission job is signed electronically, the user will have to resubmit the affected Sample(s), i.e., the one(s) with errors, with a new Sample ID (or IDs) and as a new Sample Job. However, there is a planned enhancement to allow labs and PWSs to change the status of a certified Job, make corrections, and resubmit to the primacy agency.
What to do if you cannot get State CROMERR certification?
The CMDP application is CROMERR compliant because it uses OEI’s Shared CROMERR Services (SCS) for user management, registration and e-signature. EPA has determined that if the state has an existing Attorney General (AG) certification on file with the Agency, that certification will be interpreted to apply to drinking water data reported via CMDP, which dramatically simplifies the state’s CROMERR application. If the State’s AG does not support EPA’s interpretation, then the State will have to consult with OEI on steps to complete a new AG certification.
How is Lab Certification Transferred to the CMDP and Does that Affect Analyte Methods Available?
Lab certification information is often managed in SDWIS State (or another compliance database) by the primacy agency, but in some cases is managed in an entirely separate application (e.g., MS Access). As a result, the Data Portal Work Group (DPWG) that helped define CMDP requirements determined that migrating laboratory certification information would be a future enhancement. Other Lab information, such as contact, address, lab ID, lab name, etc., are transferred to the CMDP via the Data Sync Engine (DSE). There is currently no relationship between lab certification and the analytes available in the CMDP; a future enhancement could be implemented which filters analyte-method pairs by certification status.
More Clarity on When a CMDP Samples Job will not be Accepted by SDWIS State.
SDWIS State will accept jobs submitted to the CMDP that comply and pass XML Sampling data validations.
Clarify the Analyte Pairings that Exist in SDWIS State and CMDP. Publish what is Available?
The CMDP Team is currently exploring ways to publish the existing analyte-method pairings supported by the CMDP. The EPA Technical Support Center (TSC) also publishes supported analyte-method pairings.
What Happens When a Lab Does Not Define Their Methods?
All samples data should have a corresponding method associated with the sample. Currently in CMDP the method name is a federally required field, meaning there will be a validation warning in the Validations tab of the CMDP for a missing method.
What is the process for approving drinking water methods that would be allowed in SDWIS State?
Consultation with the EPA Cincinnati Technical Support Center would be required.
If a State Has Never Used Migrate to State, How Can They Acquire This?
Migrate to State is available through the SDWIS Help Desk or through ASDWA.
Configuration Options for Samples Data Validation. (Prime vs. CMDP).
The CMDP application validates lab or water-system reported samples as defined by the CMDP Sampling Data Dictionary. Sample validation in Prime is still under development.
Inactive Facility Samples? (Use Case Identification).
The use case was a lab reporting a sample via CMDP for an inactive facility. CMDP would not reject the sample associated with the inactive facility, but it might be rejected by XML Sampling/SDWIS-State, or determined to be an invalid sample by the state primacy agency’s compliance officer.
Residential sampling points (Lead and copper) Use Case: CMDP Will Not Accept Without a Predefined Sampling Point and Residential Sampling Creates a Business Case.
Sampling Point IDs and Names in CMDP are copies of equivalent data elements migrated directly from the primacy agency’s SDWIS/State tables (or other compliance database) via the Data Synchronization Engine (DSE). For samples collected from new sampling points not yet in CMDP, in addition to informing the primacy agency directly, a PWS CMDP Administrator could use the CMDP’s Profile Change Request feature to notify the state of a new sampling point. The primacy agency would then add this sampling point information to SDWIS/State, and the DSE would automatically migrate the new Sampling Point ID (and other information) at the next scheduled run (CRON job).
Clarify SCS & CMDP authentication. How can SCS be used to administer CMDP users?
The CMDP application uses Shared CROMERR Services (SCS) for user authentication. The CMDP application does not have independent authentication. SCS administers the roles in CMDP and are described in the SCS Role Registration User Guide. More information on SCS Administration for CMDP Administrator roles can be found in the Shared CROMERR Services Advanced Help Desk User Guide located on the CMDP Help Center.
Action Items:
- Communicate definitive requirements for primacy agencies to add analyte pairings and how to add after transition.
- Composite sampling already added to Jira.
- Provide training on SCS use
RTCR Training (Monday, 5/8 pm)
Feedback & Discussion:
- Experiencing many issues due to multiple triggers. RESPONSE: EPA will conduct the following training that addresses this issue:

- DEI raised some conflicts with previous guidance. RESPONSE: In these situations, the rule is the decider. Any identified conflicts in the RTCR DEI should be sent to [enter Infrastructure lead]. EPA will address these conflicts accordingly (e.g., directly to the stakeholders; revisions to DEI).
- NC has developed a dashboard that helps them manage samples that trigger RTCR actions and keep field and HQ staff coordinated.
EPA has any estimate on reporting burden for states. RESPONSE: EPA addressed the economic and workload burden of RTCR requirements in the Economic Analysis for the Final Revised Total Coliform Rule [EPA 815-R-12-004]. This document can be found at https://www.regulations.gov/document?D=EPA-HQ-OW-2008-0878-0275.
Action Items:
Update DEI example/exhibit on page 32.
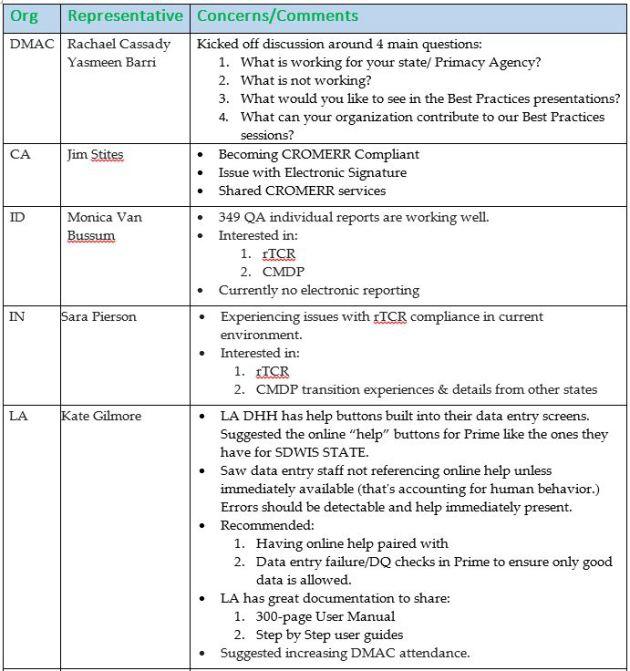
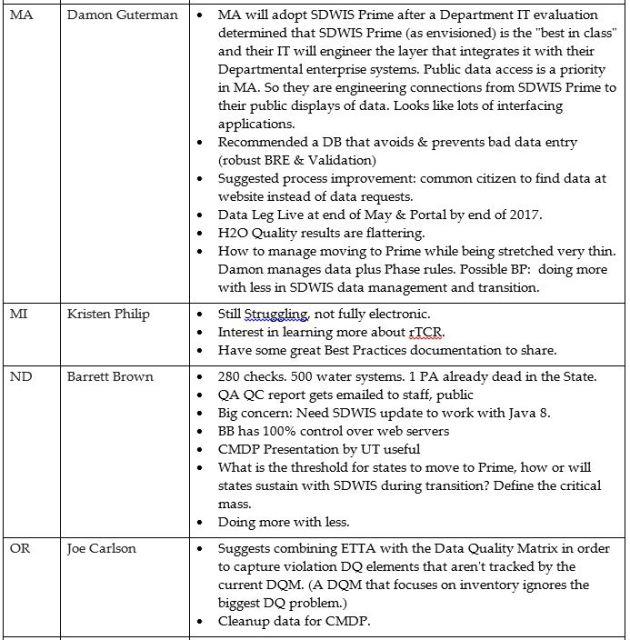
DMAC Meeting (Tuesday, 5/9 8:30 am)
Feedback & Discussion:
- Best practices are being collected by the DMAC and shared on their calls, with recordings that can be shared.
- Members expressed needs to learn about best practices in many areas of implementation but also volunteered to share best practices of their own:

IAWG Meeting (Tuesday, 5/9 am)
Feedback & Discussion:
- Discussions on use of IAWG SharePoint site and improving the methodology for gathering user stories and priorities
- Needs for a test environment and data migration tool(s) for testing data exchange solutions and associated schedule
- Discussion on the need / priority level for tool(s) that can help primacy agencies create XML files for bulk data transfer
- Need to alter our priority scheme…move away from High/Med/Low
SIWG Meeting (Tuesday, 5/9 am)
Feedback & Discussion:
- Overall agreement on approach of doing Primacy Agency samples management in SDWIS Prime and not using CMDP for this need
- Need to make crystal clear that this discussion is only around Primacy Agency samples data management – this has nothing to do with changing or touching CMDP for labs and utilities
- Disagreement among the states regarding the use of the CMDP validation rules – some say you need the same validation rules for states/primacies that are in CMDP for labs/utilities
- Definition of “minimum viable record”
- What will be configurable for states
- Conditionally required fields
- Data validation rules vs CROMERR validation protocol
- Error reporting needs to specify line item
- What sample data is editable? Lab transfer vs PA entry
Actions Items:
- Attain to document assumptions of data validation rules reuse within LOE for samples data management in Prime; estimate impact if we used different validation rules
- Follow up with SIWG members for looking more specifically at question of data validation rules
- Will follow up with product owners and SIWG regarding LOE and how to prioritize development and functionality
- CMDP Attain Support
- Attain ramping up to new level of almost doubled monthly support
- Deric Teasley and Chris Durham (Attain) working on a process for a week by week projected look ahead of when Attain will be able to provide technical support to specific Primacy Agencies needing help beyond EPA Team help
- Two tiered Attain support
- More intense (higher hours) fast track transition support
- Longer term (one to few hours) transition support
Improving Operations with Current Systems (Tuesday, 5/9 pm)
Feedback & Discussion:
- There are a number of steps for data between state databases and ETT/ECHO. The time lag contributes to many of the common problems with the data.
- Not including an implicit Return to Compliance also creates problems for data from some states. OECA is not comfortable making those decisions when they don’t have all the information they need.
- The top 10 reporting errors come from three common problems – data duplication, referential integrity, missing required data. There are recommendations for how or resolve these common data errors.
Actions Items:
- DMAC needs to reconcile this with the Data Quality Matrix.
- Share with the community the % common errors relative to total data submitted.
- We need to evaluate how these many errors will impact migration to Prime.
- Identify actions in the draft Data Quality Improvement Plan (DMAC input over the next few months) to help improvement.
- Share further info on Java resolution on SDWIS State
Getting to Know SDWIS Prime Part 1 (Wednesday, 5/10 am)
Feedback & Discussion:
- New Prime edit checks, impact on data migration
- Anticipation that Prime v1.0 may not provide all needed functionality, with primacy agency needing to supplement
- Interest in within application help, thinking of links from points in application to data dictionary
- Need for bulk violations processing, candidate schedules filtering – dashboards
Actions Items:
- Determine and communicate plan, capacity and approach for getting PA data into Prime for the 0.8 and subsequent releases so that PAs can effectively do testing as well as gap analysis for what is in Prime versus what is in current system.
- Examples include migrating data from the SDWIS Fed Warehouse, levering the current DSE tools for inventory/LE, taking full Oracle dump and migrating in
Getting to Know SDWIS Prime Part 2 (Wednesday, 5/10 am)
BRE Feedback & Discussion:
- Lag period flexibility
- Compliance on demand – pre compliance reports dashboard (i.e. compliance “report” vs. BRE “decision”)
- Black box vetting by SMEs – expect more issues identified in community testing
Using Prime to Support Program Implementation (Wednesday, 5/10 pm)
Interfacing Applications / Data Exchange Solutions Feedback & Discussion:
- Bulk upload – simple XML prep tools and/or upload format
- Concerns on APIs -> should change very little, Interfacing Applications only need to send data requests to APIs (PAs don’t need to create APIs), what programming languages use APIs
- Local data mart – most likely match SDWIS State schema, EPA pays for development, States might need to re-create additional objects (i.e. database views) that they have built from S/S.
- Versions of Prime for testing, not needed much with APIs remaining stable
Transition Feedback & Discussion:
- Clarification that use of the BPA is not required to transition to Prime, primacy agencies can use their in house and/or primacy agency specific contractor to transition to Prime
- Question on if BPA vendors can handle large numbers of primacy agencies transitioning over same time period -> vendor’s bids on contracts are designed to be scalable, EPA will hold vendors accountable for delivering transition support
- First BPA award (NH) using DWSRF set aside funds (10% “State Program Management” and 15% “Local Assistance and Other State Programs” set asides).
- Exchange Network Grants
- 8 outstanding grants
- FY17 grant awards
- Training for state administrators, other primacy agency training – videos, webinars, PA or Region specific training
e-Reporting
Feedback & Discussion:
- It saves the state time and effort and significantly reduces data errors
- Start with one lab and others will notice
- E-reporting = only way to deal with short turn around (like for lead)
- Labs will have lots of growing pains – someone at the state available for help is important
- States with existing e-reporting systems may not move to CMDP
General
- Development Decisions
- Decision tree – Inclusion of SIWG/States in high priority what’s in and what’s out decisions
- What is the plan for future Prime upgrades
- Transparency across IAWG and Prime
- Ensure Bug fixes are not cataloged as Enhancements
- How and when will state specific data be available to test with? Need for Sept release
- What is training plan for State staff?
- Incentives to move to Prime
- Dashboard
- Current to all rules
- Designed to include WIIN requirements
- Expected Lead / Copper requirements
- Clock is running (SDWIS State is dying)
- Create Transition help packet
- Can Prime become a “data lake” (or other technical option) to reduce primacy agencies’ public record request response burden?

